World’s first anode-free sodium solid-state battery
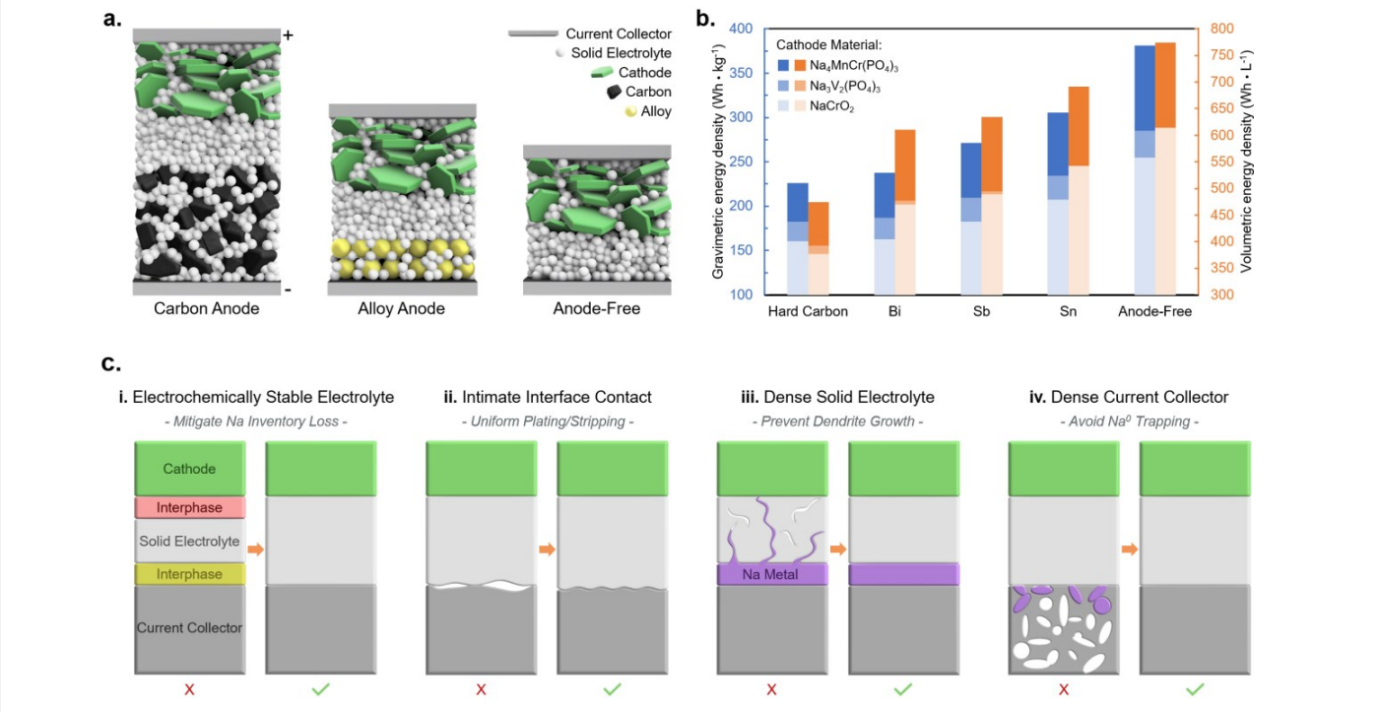
In what is described as the world first, researchers at the Laboratory for Energy Storage and Conversion (LESC) in the U.S. have managed to devise design principles for enabling an anode-free all-solid-state battery.
The LESC is a collaboration between the University of Chicago Pritzker School of Molecular Engineering and the University of California San Diego’s Aiiso Yufeng Li Family Department of Chemical and Nano Engineering.
“Although there have been previous sodium, solid-state, and anode-free batteries, no one has been able to successfully combine these three ideas until now,” said UC San Diego PhD candidate Grayson Deysher, the first author of a new paper outlining the team’s work.
To create a sodium battery, which is said to boast an energy density on par with lithium ion batteries, the research team needed to invent a new sodium battery architecture.
It opted for an anode-free battery design, which removes the anode and stores the ions on an electrochemical deposition of alkali metal directly on the current collector. Eliminating the anode enables reduced weight and volume, higher cell voltage, lower cell cost, and increased energy density, but brings its own challenges.
“In any anode-free battery there needs to be good contact between the electrolyte and the current collector,” Deysher said. “This is typically very easy when using a liquid electrolyte, as the liquid can flow everywhere and wet every surface. A solid electrolyte cannot do this.”
However, the liquid electrolytes create a buildup called solid electrolyte interphase while steadily consuming the active materials, reducing the battery’s lifetime.
The UC San Diego team took a novel approach to this problem. Rather than using an electrolyte that surrounds the current collector, they created a current collector that surrounds the electrolyte.
They created their current collector out of aluminum powder, a solid that can flow like a liquid.
“An aluminium current collector is found to achieve intimate solid–solid contact with the solid electrolyte, which allows highly reversible sodium plating and stripping at both high areal capacities and current densities, previously unobtainable with conventional aluminium foil,” the researchers reported.
During battery assembly, the powder was densified under high pressure to form a solid current collector while maintaining a liquid-like contact with the electrolyte, enabling the low-cost and high-efficiency cycling. The researchers reported that a sodium anode-free all-solid-state battery full cell has demonstrated stable cycling for several hundred cycles.
“Sodium solid-state batteries are usually seen as a far-off-in-the-future technology, but we hope that this paper can invigorate more push into the sodium area by demonstrating that it can indeed work well, even better than the lithium version in some cases,” Deysher said.
The researchers have filed a patent application for their work through UC San Diego’s Office of Innovation and Commercialization. Their findings are further discussed in “Design principles for enabling an anode-free sodium all-solid-state battery,” published in Nature Energy.















